 
Within just ten years, three elements were discovered that fit the predictions of Mendeleev (gallium, scandium and germanium) and the scientific world surrendered at his feet. His predictions were based on observing that the properties of the elements were repeated periodically in each row, hence the name Periodic Table. It wasn’t that he was practicing card reading for chemistry. He changed the position of elements that didn’t fit well because of their weight, and he left gaps open for undiscovered elements, for which he predicted their properties and atomic weights. The German chemist Lothar Meyer had done the same -without either of them knowing of the other’s work- but Mendeleev finished a year earlier, in 1869, and was more daring than Meyer. Then he realized that he could combine the two rules and, with his deck of cards of the 63 elements that were known at that time, he did something similar to the game solitaire, with the atomic weight increasing in each row and the elements with similar properties lined up in columns. Another possibility was to group them with cards of similar elements. First, he put the elements in order of the weight of their atoms. 
He wrote the data for each element on a card and locked himself in his office to arrange them. Monument to the periodic table in Bratislava, Slovakia. On February 17, 1869, Dmitri Mendeleev cancelled decided to stay at home working on how to arrange the chemical elements in a systematic way. When he set out to write one, he also decided to solve the problem of the disorder of the elements. It was just that he gave very entertaining classes without following a textbook -partly because there was no elementary treatise on chemistry in Russian. 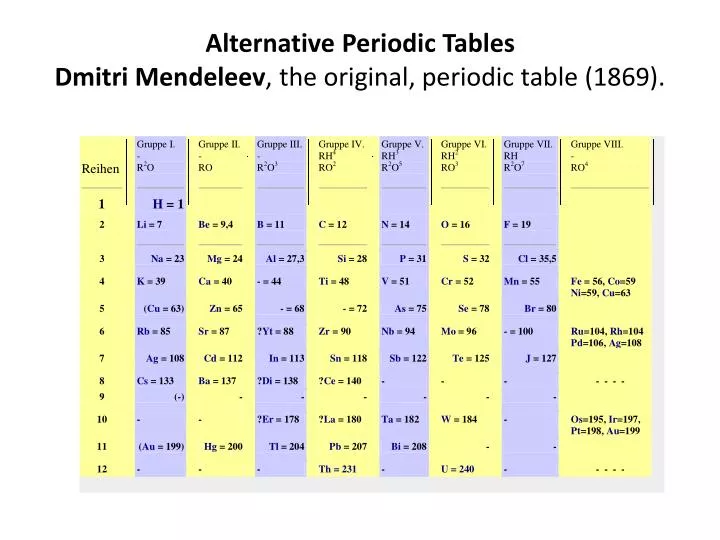
Hundreds of students flocked to hear this loud and flamboyant man, who accepted women among his students, and who only cut his beard and hair once a year. Petersburg, where Mendeleev’s lectures at the university became very popular. Both men went on to have brilliant careers in St. That same year, the writer Fyodor Dostoyevsky made the same route but in reverse: he had been deported to Siberia. His mother died shortly after fulfilling her goal and leaving Mendeleev enrolled at school. They had to cross Russia to get there, more than 6,000 kilometers from his native Siberia, which had not yet been reached by the railroad, so they hitchhiked. Petersburg so he could continue his studies. 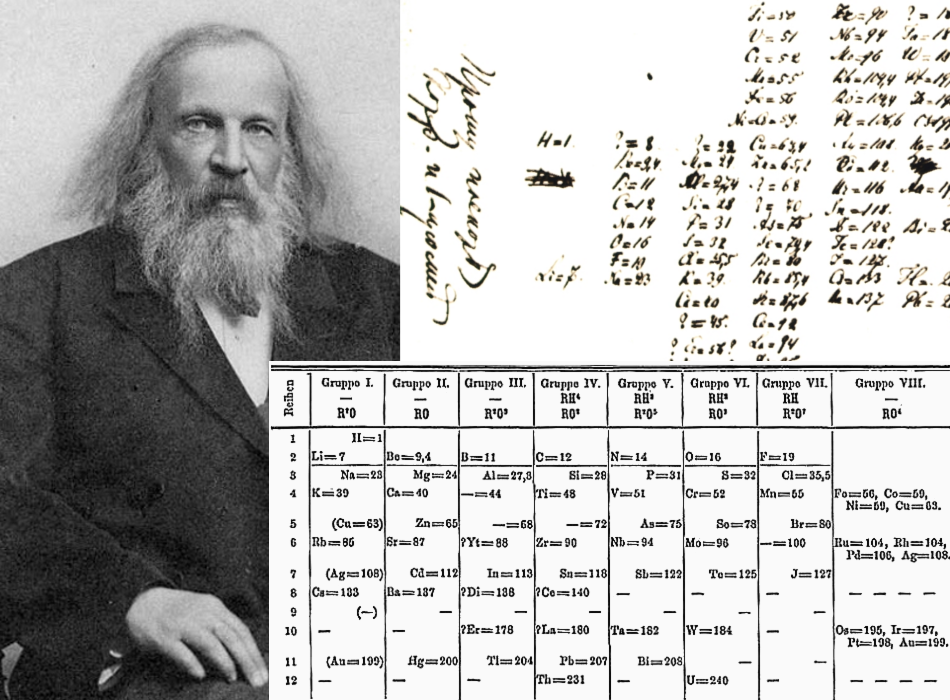
In 1849, when he was 15, his family lost everything in a fire and his mother decided to take him to St. Mendeleev did not find it easy to succeed in science. Long before that, it was already known that if one day we were able to collect trillions of atoms of copernicium, it would form a metal, since this element had a place reserved for it just below zinc, cadmium and mercury in the Periodic Table -ever since it was created in the nineteenth century by Dmitri Mendeleev ( 8 th February 1834 – 2 nd February 1907), a Russian chemist also famous because his name appears on millions of bottles of vodka. One of the last elements to officially receive them was number 112, named copernicium (Cn) in February 2010 in honor of astronomer Nicolaus Copernicus, fourteen years after German scientists created a single atom of this artificial and extremely radioactive element, which disintegrated before it could be studied carefully. The last row of the periodic table has just been completed, with the announcement of four new elements (with the numbers 113, 115, 117 and 118), although they don’t yet have names or symbols. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
